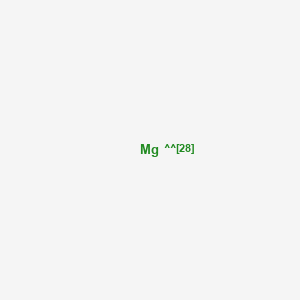PDF) Magnesium Stable Isotope Fractionation on a Cellular Level Explored by Cyanobacteria and Black Fungi with Implications for Higher Plants
Magnesium (Mg). Diagramm der atomaren Zusammensetzung und Elektronenkonfiguration eines Atoms von Magnesium-24 (Ordnungszahl: 12), die häufigste Isotop Stockfotografie - Alamy

PDF) Magnesium and carbon isotope fractionation during hydrated Mg-carbonate mineral phase transformations

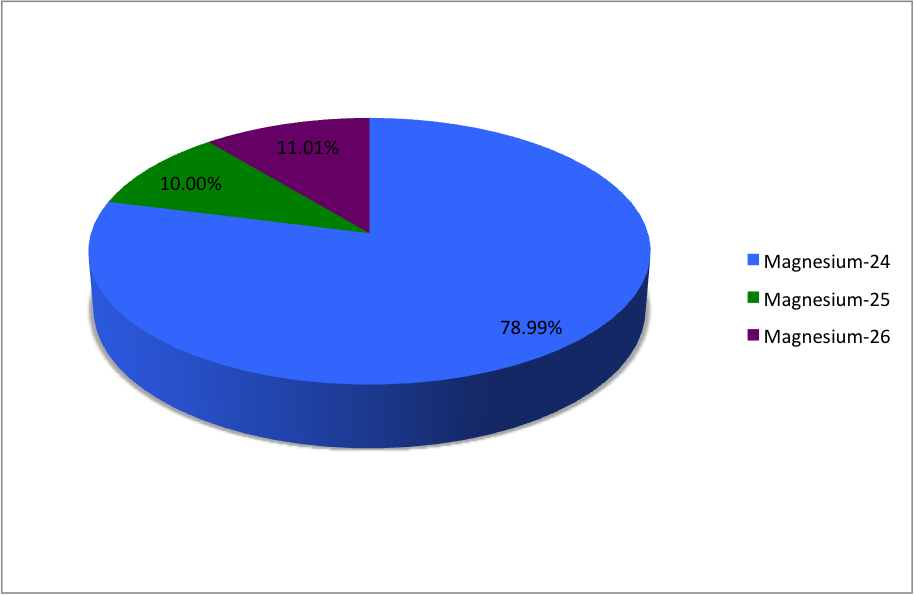
In a periodic table the average atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance on earth. The three isotopes and their
a) 12 Mg and Mg are symbols of two isotopes of magnesium. Compare atoms of these isotopes with respect to: (i) composition of their nuclei. (ii) electronic configuration and valency - CBSE

Oxygen and magnesium mass-independent isotopic fractionation induced by chemical reactions in plasma | PNAS

Illustration Av Magnesiumisotopernas Atomstruktur Stock Illustrationer - Illustration av schema, isotoper: 214177006
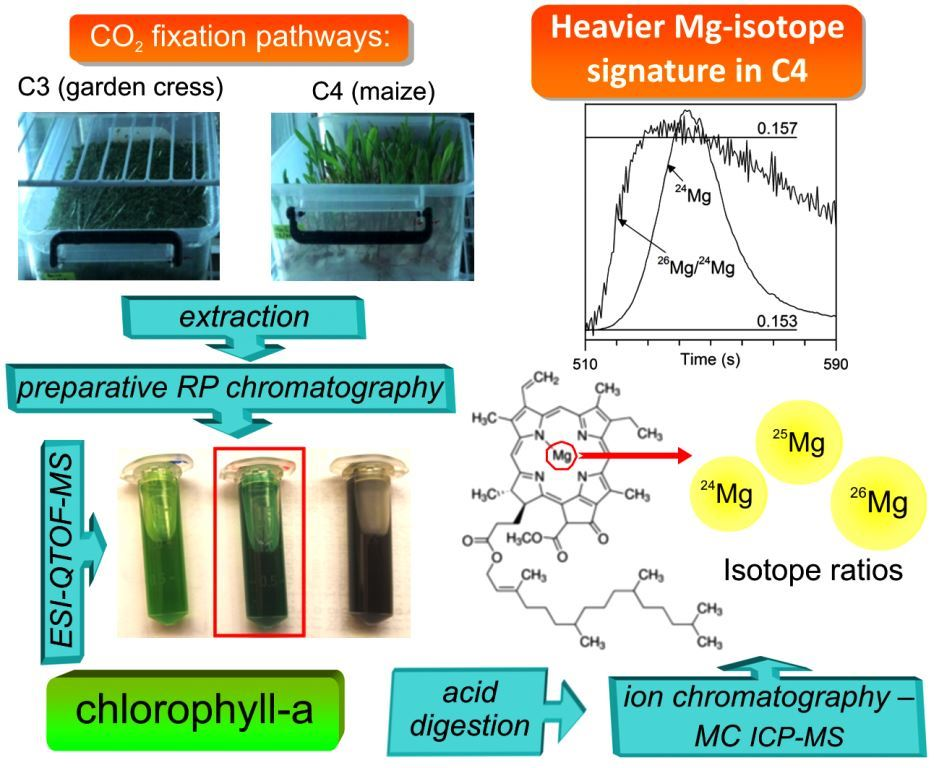
Molecules | Free Full-Text | Magnesium–Isotope Fractionation in Chlorophyll-a Extracted from Two Plants with Different Pathways of Carbon Fixation (C3, C4)

Magnesium (Z=12) has isotopes that range form Mg-20, "to"Mg-31, "Only" Mg-24, Mg-25,andMg-26 are not radioactive. What mode of radioactive deacy would convert. What mode of radioavtive deacy would convert Mg-20, Mg-21, Mg-22,