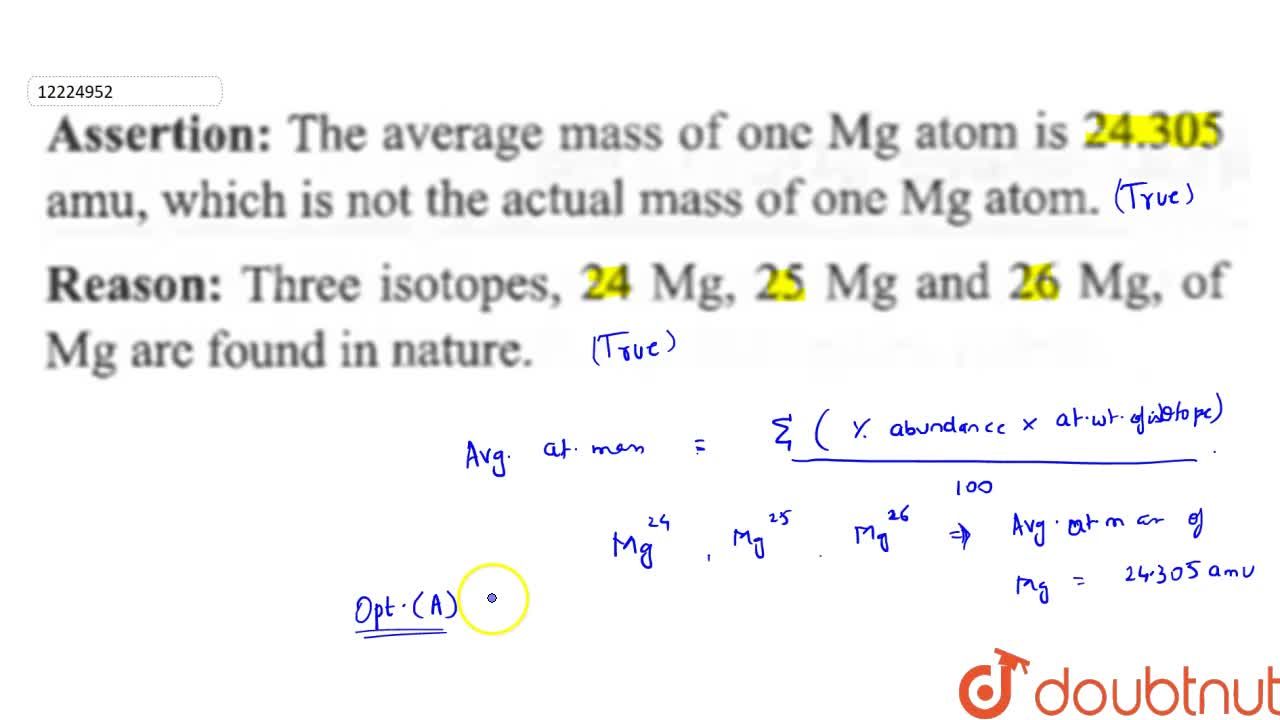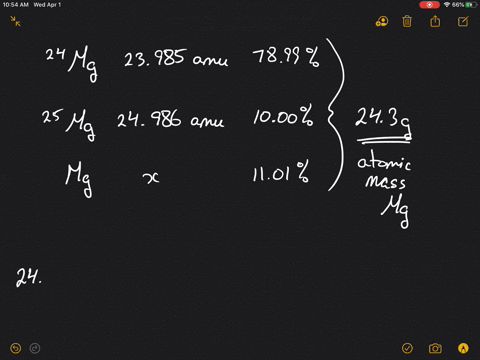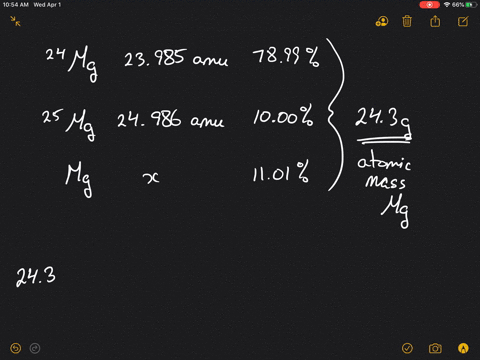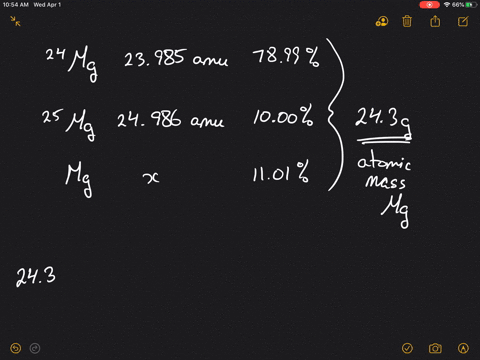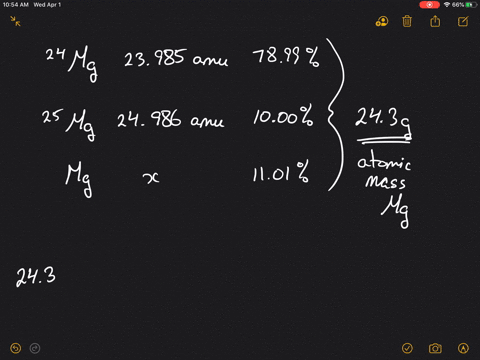
SOLVED:Magnesium has three naturally occurring isotopes: ^24 Mg (23.985 amu) with 78.99% abundance, ^{25} \mathrm{Mg} (24.986 amu) with 10.00% abundance, and a third with 11.01% abundance. Look up the atomic mass of

Magnesium has two isotopes ^2412Mg and ^2612Mg . According to which isotopes of magnesium have different mass numbers.

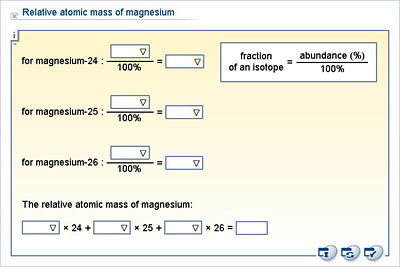
SOLVED: Calculate the average atomic mass of magnesium using the following data for threc magnesium Isotopes Ksotope massl relativg abuudance Mg-24 23.985 0.7870 Mg-25 24.986 0.1013 Mg-26 25,983 0.1417 Calculatc the average

2. Using the following data, calculate the average atomic mass of magnesium (give your answer to the - Brainly.com

Average atomic mass of magnesium is `24.31`amu. This magnesium is composed of 79 mole % of `24mg... - YouTube

Magnesium and oxygen combine in the ratio of 3:2 by mass to form magnesium oxide. What mass of oxygen gas would be required to react completely with 24 g of magnesium?

1 Warm Up Isotopes Mass of Isotope Abundance 24 Mg =24.0 amu 78.70% 25 Mg = 25.0 amu 10.13% 26 Mg = 26.0 amu 11.17% Calculate the mass average of magnesium. - ppt download

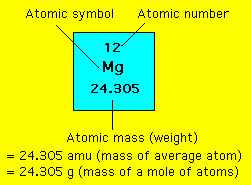
In a periodic table the average atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance on earth. The three isotopes and their